Traditional batik was made with a copper stamp or drawn on cloth by hand using a tool called a chanting. A chanting is a little copper bowl with a spout attached to a wood or bamboo handle. The chanting is dipped into hot wax. The artist draws with the chanting by using the law of gravity.The wax to pours out of the spout and penetrates the fabric.
Traditional batik was made with a copper stamp or drawn on cloth by hand using a tool called a chanting. A chanting is a little copper bowl with a spout attached to a wood or bamboo handle. The chanting is dipped into hot wax. The artist draws with the chanting by using the law of gravity.The wax to pours out of the spout and penetrates the fabric. They draw with washable markers so the original lines will disappear once the piece is waxed. They usually work on fine rayon, although batik is often done on silk or cotton.
They draw with washable markers so the original lines will disappear once the piece is waxed. They usually work on fine rayon, although batik is often done on silk or cotton. The final product is a beautiful, durable and washable painting of fabric. These batiks will not fade, shrink or bleed.
The final product is a beautiful, durable and washable painting of fabric. These batiks will not fade, shrink or bleed.
copper which is use to make the stamp table and plate.
 The stamping process requires special skill and is expertly practiced. After stamping the piece will be colored again, creating a luminous effect.
The stamping process requires special skill and is expertly practiced. After stamping the piece will be colored again, creating a luminous effect.
 The fabric, after being stamped, is pinched into a pattern. Several colors are applied systematically and allowed to bleed together.
The fabric, after being stamped, is pinched into a pattern. Several colors are applied systematically and allowed to bleed together.
 The batik now must be made permanent by applying fixative and boiling. Until a piece of batik is processed the colors are water soluble and can be destroyed easily. But batik process, especially commercial application does have adverse effects on the environment. Batik is all processed by hand, boiled in a forty four gallon drum, and washed out in the river.
The batik now must be made permanent by applying fixative and boiling. Until a piece of batik is processed the colors are water soluble and can be destroyed easily. But batik process, especially commercial application does have adverse effects on the environment. Batik is all processed by hand, boiled in a forty four gallon drum, and washed out in the river.
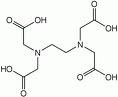
The whole Batik process revolves around the principle that wax and water repel each other. When hot liquid wax is applied to cloth, it hardens almost immediately. The wax then acts essentially as a mask, protecting the area of cloth saturated with the wax from becoming saturated with water. If dye is added to water, the area of fabric left exposed and not covered by wax becomes saturated with the dye. Dye cannot penetrate a fabric where wax is painted.

paraffin wax
It is therefore possible to build up multicoloured batik by waiting for the cloth to dry and waxing in previously dyed sections to prevent the next dye colour from taking. This principle works very effectively if work from light to dark coloured dyes. Another, quicker method is to draw an outline of hot wax onto the fabric, creating completely enclosed areas or circles of wax. Dye is then hand-painted with in the wax borders. These borders stop the different coloured dyes from running into each other.

bee wax
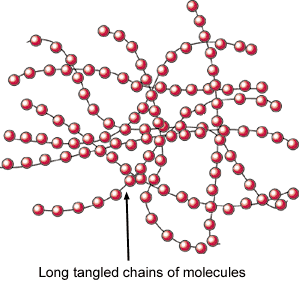
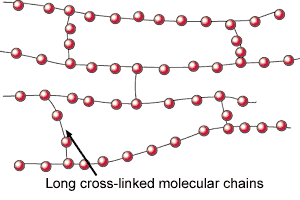
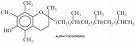
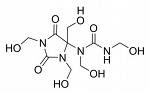
Wax is one of the oldest forms of textile resist and is perhaps the strongest. It is possible to use it in long, cool, immersion dye baths and in hand painting projects. Natural beeswax and paraffin wax can all be used in numerous combinations for a variety of effects. Natural beeswax is malleable and tacky and when mixed to a ratio of one to one with paraffin creates a strong resist and the characteristic crackle that defines batik. It may also be used at a ratio of 70% beeswax to 30% paraffin to create concise detail with little crackle.